
The Liberal west narrative has a way to equate the unthinkable. They take the understanding of how an idea works in the West colonial structures and juxtapose it with other cultures.
"Hindu Nationalists". That is supposed to evoke the idea of "German Nationalists," as if there is an equivalence.
The crimes of the Colonialists are to be plastered on the victims of colonialism. The Slave masters' jingoism is sought to have an echo in the search for the identity of those enslaved.
For the last over 1400 years, the Islamic hordes and the European savages have enslaved other human beings from Africa, Asia, North and South America, and Australia-NZ. All to prove their superiority. And that of their ideologies, prejudices, and imperialist hate narratives.
When the colonialist or imperialist asserts his superiority by emphasizing his identity, he is using it as a weapon. When the enslaved and the colonized want to find their voice by piecing together their identity that was shredded and trashed by the colonial masters, he is reaching out for survival.
The assertion of identity by the colonialist and that by the colonized can never be the same!
When a European, a land where every country carries the crime of having colonized and dismantled the identities of the ones they attacked, shouts nationalism it is an echo of that past.
When an African or a Hindu (whose culture and identity were trampled upon by the imperialists) puts his identity up, it is also an echo from the past. A past that was ruthlessly and senselessly trampled upon and attempted to be dismantled.
To ding an African or a Hindu for taking refuge in his identity of an ancestry that has suffered immeasurable and inhuman attacks is to continue the imperialist experiment.
And this in our current world is being perpetrated by those who call themselves liberal.
They are anything but.
Beneficiaries of imperialist plunder and crimes whose soul searching were selective and timed for their own profits. These are worse than those who actually committed the crimes. For, them you can isolate. These criminals who kill by proxy and profit by manufactured human rights truly take any humanity that is left on the planet to extinction.
The battle for control on the planet is not only from the abusers. But those who wear the cloak of piety while they enable the abusers.
The battle is with these self-styled moral busybodies.
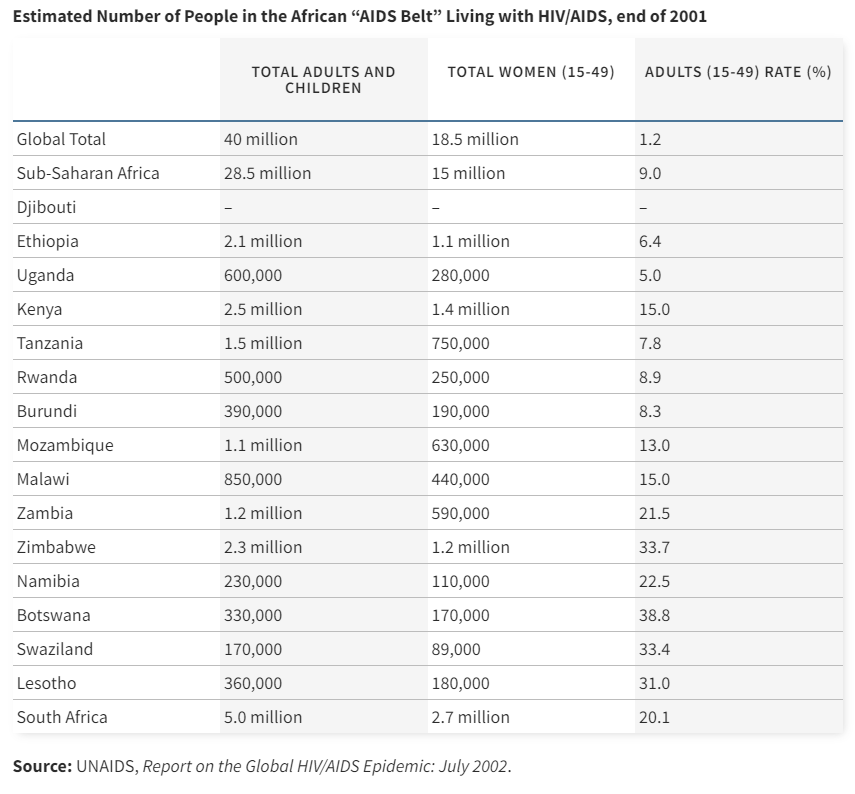
Saving the Sub-Saharan Poor
In 2000 when the poor in Sub-Saharan Africa were reeling from AIDS, the cost of drugs to fight the epidemic was between $10,000 and $15,000 per year.
SSA* is the region of the world most severely affected by HIV and AIDS. Uganda, Kenya, and Tanzania were among the countries where the HIV epidemic was first recognized during the early 1980s. In 2000, an estimated 25.3 million persons in SSA were infected with HIV, and the average national prevalence of HIV infection among persons aged 15--49 years was 8.8%. Approximately four million new infections occurred during 2000. Approximately 10% of persons aged 15--49 years are infected in 16 countries, including seven in southern and eastern Africa, where approximately 20% are infected. In Botswana, the country with the highest prevalence, 36% of the adult population is infected with HIV (Source: "The Global HIV and AIDS Epidemic, 2001"/CDC)
Sub-Saharan Africa accounted for over 70% of AIDS patients globally.
But the rich world did not care. And Pharma majors wanted their own pound of flesh for the research they did.
Health, you see, ceased to be about humanity anymore. It was about profits.
It is not that people have not done cutting-edge research and work throughout history but they did not deliberately allow people to die because even when the cure was there, the patient did not have enough means to afford it.
Like Nkosi Johnson.
At the 13th International Aids Conference in July 2000, this 12-year-old boy gave South African President Thabo Mbeki a life lesson. So much more had to be done to save the millions who were infected.
He or others in Africa did not have enough money to afford the drugs.
Nkosi died on June 1st 2001.
In August 2000, one afternoon four Americans and one Indian met discretely in an apartment in London.
To reverse the future. What was happening in Sub-Saharan Africa, was a crime against humanity.
The Indian was CIPLA CEO Yusuf Khwaja Hamied.
While the big pharma companies were trying to make profits, he was hatching the plan to save the world.

Hamied wanted to reduce the cost of the drugs to $1/day (from the $15,000 per year). In 10 years, since that meeting in London, the price had reduced to 20 cents. Over 6 million had been administered the drugs
Dr. Yusuf K. Hamied, chairman of the Indian drug giant Cipla Ltd., electrified the global health community a decade ago when he said he could produce cocktails of AIDS medicines for $1 per day — a fraction of the price charged by branded pharmaceutical companies. That price has since fallen to 20 cents per day, and more than six million people in the developing world now receive treatment, up from little more than 2,000 in 2001. (Source: "As U.N. Meets, a Battle Over Generic Biotech Drugs"/New York Times)
The dice had been cast. CIPLA was taking on Big Pharma and attacking their profits.
In 2001, 40 drug companies including the UK’s GlaxoSmithKline slapped a joint legal action on the South African government to block it from buying cheaper medicines from the Indian companies.
The protest coincides with the opening day of legal action against the South African government in Pretoria, brought by GSK and 41 other pharmaceutical companies. The 42 firms have launched a joint corporate legal challenge to the South African Medicines and Related Substances Control Amendment Act 1997, which they argue would allow the health minister to arbitrarily ignore patents on medications. (Source: Protest in Britain as drug companies sue South African government/ The Independent)
For three years, this group of pharma companies hired a battery of patent lawyers pouring in millions and trying every trick in their arsenal to deny the treatment to the African poor. In the interest of their profits, of course.
The Indian Pharma industry
India's pharma industry is a very formidable player globally.
The Indian Pharmaceuticals industry plays a prominent role in the global pharmaceuticals industry. India ranks 3rd worldwide for production by volume and 14th by value. The nation is the largest provider of generic medicines globally, occupying a 20% share in global supply by volume, and is the leading vaccine manufacturer globally. India also has the highest number of US-FDA compliant Pharma plants outside of USA and is home to more than 3,000 pharma companies with a strong network of over 10,500 manufacturing facilities as well as a highly skilled resource pool. The pharmaceutical industry in India offers 60,000 generic brands across 60 therapeutic categories. Major segments include generic drugs, OTC Medicines, API/Bulk Drugs, Vaccines, Contract Research & Manufacturing, Biosimilars and Biologics. (Source: InvestIndia)
India has the highest number of USFDA-approved manufacturing plants. The highest in the world.
India has 665 USFDA-approved manufacturing plants outside the US, the highest in the world. Inspections had stopped early last yeardue to the pandemic. In fact, several plants are also in different stages of redressal, following earlier observations made by the USFDA. (Source: USFDA resumes inspection of manufacturing plants in India/ The Hindu Business Line)
India's contribution to the American generics industry is substantial. Indian companies have the largest share.
Note for the chart below: A Drug Master File (DMF) is a submission to the Food and Drug Administration (FDA) that may be used to provide confidential detailed information about facilities, processes, or articles used in the manufacturing, processing, packaging, and storing of one or more human drugs. (Source: FDA)
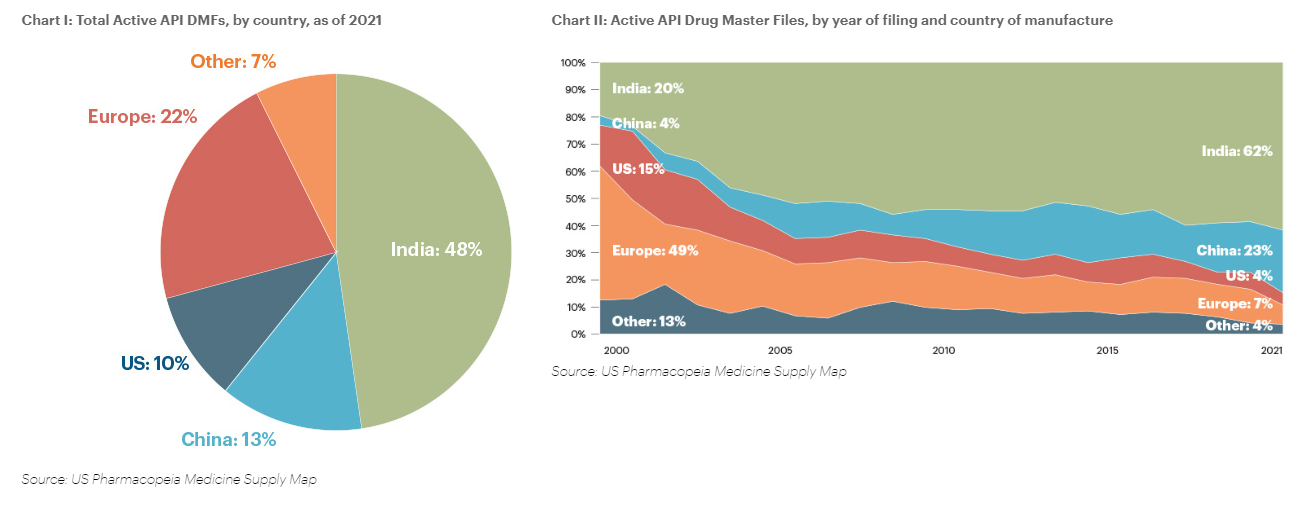
Indian pharma has come a long way because of the initial push it received from the Indian laws that enabled the companies to reverse engineer the drugs and create generic versions for the Indian customers and outside.
Get a Weekly Insightful Newsletter: If you are here via a search or because someone shared this newsletter with you, please so subscribe to some insightful content that is published every Sunday morning US time and evening India time.
Patents and the Indian Pharma industry
Patent rights came into force in 1856. In 1970 the Patent Act was passed. India was a signatory of the Paris Convention for the protection of industrial property, 1883, and the Patent Cooperation Treaty, 1970. The Patents Act of 1970 was important in what it considered to be patentable.
The Patents Act 1970 had a very limited scope of protection wherein the essential elements of invention were new, useful and manner of manufacture. Even though manufacture was not defined in the old Act, the Patent Office established the practice of interpreting manufacture as process resulting in a tangible product. (Source: Mondaq)
Significantly, pharmaceuticals and agrochemical products were excluded from the eligibility for patenting.
The result was that only the process was patentable in India. Not the product.
And, that had a major impact.
It created a whole industry in agrochemicals and pharmaceuticals on the back of reverse engineering. Drugs as products that were patentable in the "Industrialized world" were not patentable in India.
Cheaper versions of the same drugs were made available.
World Trade Organization (WTO) was established on Jan 1st, 1995.
The 8th round of the Multilateral Trade Negotiations (MTN) was called the Uruguay Round. It was conducted within the General Agreement on Tariffs and Trade (GATT) framework. These negotiations spanned from 1986 through 1993.
123 countries were signatories and "contracting parties".
The result was the World Trade Organization or WTO.
Intellectual rights became critical in the Uruguay Round. An agreement was discussed and negotiated during the Round on Trade-Related (Aspects of) Intellectual Property Rights (TRIPS).
India signed the GATT on April 15th, 1994.
That is when it was incumbent on India to maintain the provisions related to the patents for both pharmaceutical products and process inventions.
After that, this is how the legislation in India progressed. (Source: Patent Law in India)
- On 26th March 1999, the Patents (Amendment) Act, 1999 came into force with retrospective effect from 1st January 1995. Per Section 5 (2), although the applications were filed for patents in the areas of drugs, medicines, and agrochemicals, they were kept pending in a "mailbox" or "Black Box" and opened on January 1st, 2005.
- The second phase of the amendment was brought in by the Patents (Amendment) Act, of 2002. This came into force on 20th May 2003. In this amendment, the definition of "invention" was made in conformity with the provisions of TRIPS Agreement by adding the concept of inventive step.
- The third and final amendment to the Patents Act, of 1970 was brought in by the Patents (Amendment) Ordinance, of 2004. This was later replaced by The Patent (Amendment) Act, 2005, and Patents (Amendment) Rules, 2006. They were applied with retrospective effect from 1st January 2005.
With the final amendment, India finally was aligned to the obligations under TRIPS. The important provisions of the Patents (Amendment) Act 2005 related to product patents were: (Source: Indian Patents Act)
- Extension of product patent protection to products in sectors of drugs, foods and chemical.
- Term for protection of product patent shall be for 20 years.
- Introduction of a provision for enabling grant of compulsory license for export of medicines to countries which have insufficient or no manufacturing capacity; provided such importing country has either granted a compulsory license for import or by notification or otherwise allowed importation of the patented pharmaceutical products from India (in accordance with the Doha Declaration on TRIPS and Public Health)
- Section 3 (d) regarding patentability.
Even though the laws were changed, a small "loophole" was still left creating angst amongst the foreign players.
In 2005 the Indian Patents Act, 1970, which provides patent protection for pharmaceutical products and drugs, was amended. This amendment, however, only provided patentability of pharmaceutical substances to the extent that patents would apply to new chemical entities. Section 3(d) of the Patents Act, 1970, explains that a new form of a known substance, new property / new use for a known substance and the mere use of a known process are not patentable and is not considered distinct from the known substance. Based on this principle, inventions using known substances are not patentable unless, for example, the alleged invention displays a significant increase in efficacy. Though there were several outcries from both the generic pharmaceutical and other innovator companies, the fact of the day is that section 3(d) still controls patent eligibility[3]. There are still several patents that are being granted for salts, ethers, esters, polymorphs and other “derivatives of known substance” as mentioned in the clause. This is because the Indian law provides a provision for patenting such derivatives if there is an enhancement of efficacy. (Source: The Global Significance of India’s Pharmaceutical Patent Laws / American Intellectual Property Law Association)
The time and respite that the Indian pharmaceutical industry got during the transition afforded it the ability to grow, expand and add its own indigenous R&D.

The post-2005 period saw Indian pharmas investing more in research and development (R&D) to compete with global companies. In fact, some were even developing their own new molecules.
By 2019, the pharma market in India had the following components.

The growth in the Indian pharma industry has been rapid. It is expected to grow to a $130 billion market by 2030 and become the leading provider of medicines to the world.

It was this ascendency of Indian Pharma that Angela Merkel cribbed about at the height of the second wave when India was reeling and had to divert its own vaccine production for its people as opposed to exporting. (Also, please note in this report how the Delhi CM Arvind Kejriwal was breaking the rules of closed-door meetings with the PM)
Merkel arrogantly announced Europe allowed India to become "such a large pharmaceutical producer" since India was not able to send the supplies that it had to use for its own people in the worst hit days during COVID.
Currently, as Indian pharma grows, it has become the "pharmacy to the world".
Indian pharmaceutical sector supplies over 50% of global demand for various vaccines, 40% of generic demand in the US and 25% of all medicine in the UK. The domestic pharmaceutical industry includes a network of 3,000 drug companies and ~10,500 manufacturing units. India enjoys an important position in the global pharmaceuticals sector. The country also has a large pool of scientists and engineers with a potential to steer the industry ahead to greater heights. Presently, over 80% of the antiretroviral drugs used globally to combat AIDS (Acquired Immune Deficiency Syndrome) are supplied by Indian pharmaceutical firms. India is rightfully known as the "pharmacy of the world" due to the low cost and high quality of its medicines. (Source: Indian Brand Equity Foundation)
But is it all hunky dory for the Indian pharmaceutical companies?
No.
Because the Indian pharmaceutical sector is critically dependent on China for the key active pharmaceutical ingredients or APIs.
However, India’s $42 billion pharmaceutical sector is heavily dependent on China for key active pharmaceutical ingredients or API — chemicals that are responsible for the therapeutic effect of drugs. According to a government report, India imports about 68% of its APIs from China as it’s a cheaper option than manufacturing them domestically. However, an estimate by the Trade Promotion Council, a government supported organization, puts the figure of API dependence on China at about 85%. Another independent study carried out in 2021 points out that while India’s API imports from China are at nearly 70%, its dependence on China for “certain life-saving antibiotics” is around 90%. Some drugs that are highly dependent on Chinese APIs include penicillin, cephalosporins and azithromycin, the report said. (Source: "India wants to be the ‘pharmacy of the world.’ But first, it must wean itself from China"/CNBC)
But in 2020, India decided to create a $1.3 billion fund to encourage Indian companies to manufacture APIs domestically.
India will set up a nearly 100-billion-rupee ($1.3 billion) fund to encourage companies to manufacture pharmaceutical ingredients domestically after supply chain disruptions due to the coronavirus pandemic exposed the country’s dependence on China and raised the specter of drug shortages. The program includes spending on infrastructure for drug manufacturing centers, and financial incentives of up to 20% of incremental sales value over the next eight years, according to a government statement. (Source: "India to Spend $1.3 Billion to Boost Pharmaceutical Production"/Bloomberg)
The impact of these efforts would be that the import dependence on China would reduce by 25% in 2024.

Speaking of the COVID impact on Indian Pharma. Let us go into that now.
Vaccine Apartheid: Profits from Misery
Check this map of the countries where the populations have received vaccinations. What jumps out at you?
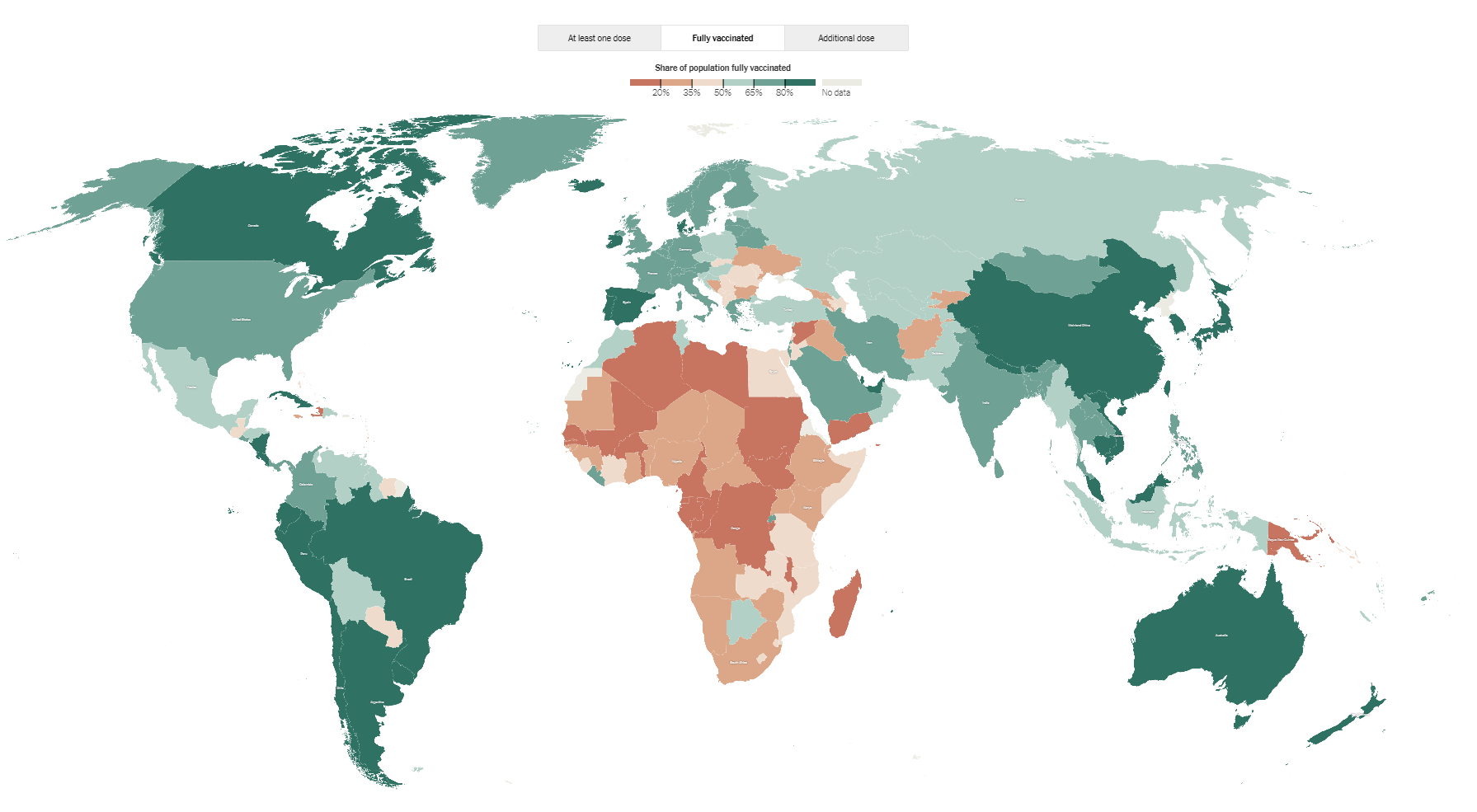
Again, just as during the AIDS/HIV epidemic in Africa, the COVID-19 pandemic saw the African continent and other poor countries losing out on vaccines that could cure because they did not have enough money or clout to get the vaccine.
Over 75% went to only 10 countries!
Roughly three-quarters of the available vaccine doses have been distributed to only 10 countries, while current projections indicate that much of the developing world will have to wait years to reach significant vaccination levels. After coming into office, Biden reversed course from the Trump administration and committed $4 billion to COVAX, the international program backed by the World Health Organization and a number of global public health nonprofits, to buy and distribute coronavirus vaccines to countries in need. But the COVAX program, which continues to face underfunding, will not be able to vaccinate 90 percent of people in 67 low-income countries this year. Some developing nations may have to wait until 2024 to achieve adequate levels of vaccination. (Source: The Intercept)
Even though the world needed it, the US and European drug companies did not allow the patent waiver to come in place. That could have allowed other generic manufacturers to produce the vaccine cheaply for the Global South.
It never happened. Most significantly because the pharmaceuticals industry had its fangs deeply embedded within the Biden administration. Those who stood to profit from the COVID vaccine companies - Moderna, BioNTech, and Pfizer - were running the show, if you will.
How Biden's administration is closely linked to the pharmaceutical companies.
Anita Dunn: White House adviser
Dunn’s financial disclosures, first reported by CNBC’s Brian Schwartz, reveal that the senior Biden adviser has consulted for several major corporations with business before the government, even though the firm has long emphasized that it neither lobbies the federal government nor represents clients on matters before the government. In the past two years, Dunn advised AT&T, Lyft, Pfizer, Micron, Intra-Cellular Therapies, Salesforce and Reddit, the disclosures show. (Source: Politico)
White House adviser Anita Dunn is co-founder of the consulting firm SKDK, which works closely with Pfizer; Biden’s domestic policy adviser, Susan Rice, holds up to $5 million in Johnson & Johnson shares; and White House science adviser Eric Lander holds up to $1 million in shares of BioNTech, which co-developed Pfizer’s coronavirus vaccine.
Dr. Eric Lander: White House science adviser

Secretary of State Anthony Blinken
He has consulted for Gilead Science. The company that produced remdesivir. Remdesivir was the only COVID-19 treatment approved by FDA.
Chiquita Brooks-LaSure: Admn for Centers for Medicare & Medicaid Services
She is the administrator of the Centers for Medicare & Medicaid Services in the Biden administration since May 2021. Was the attorney for both Pfizer and Gilead on federal policy issues. (Source: Public Financial Disclosure)
What did this conflict of interest situation lead to?
Immoral Profits funded by the American Taxpayers
The revenue and profits windfall for the three pharma majors - Moderna, BioNTech, and Pfizer - was unprecedented in the last two years.

But all this was done not by investments that these pharma companies made themselves.
They were funded by the American taxpayer instead. Interestingly named Operation Warp Speed (OWS), the COVID-19 relief program gave doles to the tune of $22 billion to Big Pharma. (Source: How The Covid-19 Vaccine Injected Billions Into Big Pharma—And Made Its Executives Very Rich/ Forbes)
Here are some specifics of who got how much.
According to a study in the leading journal Health Affairs, “The government essentially removed the bulk of traditional industry risks related to vaccine development: a) scientific failures, b) failures to demonstrate safety and efficacy, c) manufacturing risks; and d) market risks related to low demand.” The federal government spent between $18 billion and $23 billion to develop all the COVID vaccines, various estimates show. The study’s authors found that amount was given “on top of years of government-funded vaccine research that set the platforms for the current successes.” In the case of Moderna, federal agencies funded 100% of the $2.5 billion it spent developing its COVID vaccine. (Source: "How Big Pharma profited from the pandemic"/Philadelphia Inquirer)
The American taxpayers funded the research, while the profits were made by the pharma companies' executives and their political cronies.
Are Quality Standards the new weapon?
Let us first understand the two different sets of quality standards that impact Indian Pharmaceutical companies.
The first one is the Current Good Manufacturing Practice (CGMP) from the FDA.

The Indian legal framework is defined under Schedule M of the Drug and Cosmetic Act, of 1940. Good Manufacturing Practice (GMP) is defined there.

In May 2013, the Indian pharmaceutical major Ranbaxy was charged with false claims and manufacturing and distributing adulterated drugs.
Note: Under the FDCA, a drug is adulterated if the methods used in, or the facilities or controls used for, its manufacturing, processing, packing, or holding do not conform to, or are not operated or administered in conformity with, current Good Manufacturing Practice (cGMP) regulations.

In October this year, 69 children died in Gambia and the WHO announced that it was all due to the cough syrups manufactured in India.
WHO Director-General Tedros Adhanom Ghebreyesus said, “WHO has issued a medical product alert for four contaminated medicines identified in The Gambia that have been potentially linked to acute kidney injuries and deaths among children." The four medicines are cough and cold syrups produced in India, said the WHO statement. (Source: LiveMint)
The culprit company was Maiden Pharmaceuticals and its Sonipat plant. Or so the WHO alleged.
Apparently without enough evidence.
But that did not stop the global press to link the deaths, without evidence to the Indian cough syrups.
The damage was done.
The Indian government committee, however, investigated the claims and found the clinical data shared by WHO was inadequate.
The government-appointed committee investigating claims that four cough syrups made in India may have led to the deaths of 66 children in The Gambia has found that the clinical data provided by the WHO so far is insufficient to identify the aetiology. (Source: "India committee tells WHO: Information on 'killer' cough syrups 'inadequate'" / WION)
The Gambian investigators also did not see a linkage between the deaths and Indian products. For one, other kids who had died in a similar manner had not taken the Indian syrups.
The small West African state has been investigating a mysterious slew of child deaths in recent months, which police said in a preliminary investigation was linked to four cough syrups made in India. World Health Organization (WHO) investigators have found "unacceptable" levels of diethylene glycol and ethylene glycol in the products, which were made by New Delhi-based Maiden Pharmaceuticals Ltd. But Gambia's Medicines Control Agency, a national regulatory body, has not yet pinpointed the exact cause of the deaths, said Tijan Jallow, an officer at the agency. "We haven't concluded yet it is the medicine that caused it. A good number of kids died without taking any medications," Jallow told a news conference. "Other kids died, the medication that they took, we have tested them and they are good," he added. The agency is trying to establish exactly which medications, if any, each child took. (Source: "Gambia says it has not yet confirmed cough syrup as cause of child deaths"/Reuters)
So, here is the rub.
- WHO's Director General was proclaiming that Indian drugs were the reason for the deaths of the kids in Gambia in the very first week of October.
- The Gambian investigators were not certain, given the evidence, that it was indeed the Indian products that caused the deaths. As late as October 31st!
Why exactly did the Director General of WHO so eager to put the Indian pharmaceutical company in the dock? Without any evidence or investigation?
In diplomatic circles, WHO Director-General Tedros Adhanom Ghebreyesus is not necessarily known for his integrity and how he has kept his organization impartial.
In fact, he was seen as a prejudiced official of an agency that failed the poor countries during COVID and helped the rich ones to get ahead.
He has been called Dr. Cover-up (Source: Dr Cover-up: Tedros Adhanom’s controversial journey to the WHO/ORFonline)
WHO was also playing the geopolitics game against India on at least two occasions.
- One, when the COVID-19 toll was being calculated. While the Indian government estimates were 520,000, the WHO was hell-bent on giving out figures for COVID mortality at over 4 million.
- Second, WHO suspended the supply of India-made Covaxin through UN procurement agencies, and recommended that countries using the vaccine take action “as appropriate”.
How the US and the European countries hindered and hurt the Indian vaccine production as well as the response has been documented by us in our earlier newsletter here.

Close on the heels of the Gambia WHO scandal, the FDA has sent a warning to Lupin Ltd. about its Tarapur plant They have accused Lupin of "significant deviations from current good manufacturing practice (CGMP) for active pharmaceutical ingredients (API)." (Source: FDA) Main deviations are:
- Violations of poor manufacturing practices
- Problems with claims for what a product can do
- Incorrect directions for use.
FDA had withheld new approvals and listings for Lupin.
Also, check this thread on Twitter.

Indian Pharma paying for Target India campaign by US and EU?
In a very interesting article, on Fierce Pharma, Kevin Dunleavy says that Indian pharma is paying for India's hesitance to condemn Russia.
As India’s prime minister Narendra Modi faces increasing pressure from the west to condemn Russia, the country’s booming pharma industry also has come under scrutiny. Many pharma companies in India do extensive business with Russia and Ukraine. Last fiscal year, for example, India exported more than $591 million worth of pharma goods to Russia and $181 million to Ukraine, Business Standard reports. Those figures represented growth rates of 7% and 44% respectively from year to year. (Source: "India's hesitance to condemn Russia puts the country's pharma industry in compromising position" / Fierce Pharma)
The seems to be a sense that the USFDA is intentionally targeting the Indian Pharma companies - whether due to the lobbying by the US and European pharma giants or out of geopolitical compulsions.
In fact, the US Food and Drug Administration (FDA) Commissioner Margaret Hamburg was forced to deny this. Which essentially means it was something that is being seriously talked about in many circles.
The United States is “not targeting” Indian companies, but has a strict quality control regime for all products being imported into America, the head of the US Food and Drug Administration said today. “We are not targeting Indian companies. We are undertaking our required regulatory activities. We inspect and take appropriate action against companies within the United States,” US Food and Drug Administration (FDA) Commissioner Margaret Hamburg told reporters during a conference call. (Source: "US ‘not targeting’ Indian pharma companies, says FDA chief"/Indian Express)
With so much at stake - ungodly profits on the back of American taxpayers and global domination over the health and wellbeing of people, rich people actually - the battles will be fierce in future in the pharmaceutical industry.
The key will be for generic pharma manufacturers to create alternatives for the poor and save them when epidemics and pandemics hit again in future. And, no one is willing to care about them at all. Yet again.
RAW RESEARCH
Here are the documentation and links that I saved as part of my research for an article. Many of these links I may have shared in my article and many I would have used in understanding the topics at hand. Beyond this newsletter, I tend to collate all my research even in the future into these sub-pages. So bookmark them for future reference. Please feel free to dive in and use it for your own analysis.

Get a Weekly Insightful Newsleter: If you are here via a search or because someone shared this newsletter with you, please so subscribe to some insightful content that is published every Sunday morning US time and evening India time.
Video Corner: The Forest Man
Jadav "Molai" Payeng is an environmental activist and forestry worker from Majuli who has planted around 40 million trees to create a real man-made forest in the last 30 years! He is popularly known as the Forest Man of India. He has transformed the barren sandbar of the Brahmaputra into a forest.
This is his remarkable story. Worth watching!
If you like our content and value the work that we are doing, please do consider contributing to our expenses. CHOOSE THE USD EQUIVALENT AMOUNT you are comfortable with.
If you like this post - please share it with someone who will appreciate the information shared in this edition.
Today’s ONLINE PAPER: Check out today’s “The Drishtikone Daily” edition. - THE DRISHTIKONE DAILY












